No language detected.
Please check the input language, no language detected.
Please check the input language.

[Daily Farm] Last year, 1,488 drugs were approved…30% of diabetes treatments [Daily Farm = Reporter Lee Hye-kyung] A total of 1,488 medicines were found to have been approved by the Ministry of Food and Drug Safety last year. The Ministry of Food and Drug Safety announced on the 30th that it has published a report on the permission of medicines, quasi-drugs, and medical devices containing the current status of permits & 8231; certification & 8231; and reports in 2023. In the case of pharmaceuticals, a total of 1,488 items were approved, including 1,300 finished medicines, 49 raw materials medicines, and 139 herbal medicines. For finished medicines, 884 specialized medicines (68%) and 416 general medicines are empty…www.dailypharm.com [Daily Farm] Last year, 1,488 drugs were approved…30% of diabetes treatments [Daily Farm = Reporter Lee Hye-kyung] A total of 1,488 medicines were found to have been approved by the Ministry of Food and Drug Safety last year. The Ministry of Food and Drug Safety announced on the 30th that it has published a report on the permission of medicines, quasi-drugs, and medical devices containing the current status of permits & 8231; certification & 8231; and reports in 2023. In the case of pharmaceuticals, a total of 1,488 items were approved, including 1,300 finished medicines, 49 raw materials medicines, and 139 herbal medicines. In the case of finished medicines, 884 items (68%) of specialized medicines and 416 items of general medicines are empty… www.dailypharm.com

[Daily Farm = Reporter Lee Hye-kyung] A total of 1,488 medicines were found to have been approved by the Ministry of Food and Drug Safety last year. The Ministry of Food and Drug Safety announced on the 30th that it has published a report on the permission of medicines, quasi-drugs, and medical devices containing the current status of permission, certification, and reporting in 2023. In the case of pharmaceuticals, a total of 1,488 items were approved, including 1,300 finished medicines, 49 raw materials medicines, and 139 herbal medicines. In the case of finished drugs, 884 specialized drugs (68%) and 416 general drugs were approved. Last year, as the permission for rare drugs continued to increase, there were many permissions for diabetes treatments among all finished drugs. However, since the implementation of the 1+3 system, the permission for generic drugs has been steadily decreasing. [Daily Farm = Reporter Lee Hye-kyung] A total of 1,488 medicines were found to have been approved by the Ministry of Food and Drug Safety last year. The Ministry of Food and Drug Safety announced on the 30th that it has published a report on the permission of medicines, quasi-drugs, and medical devices containing the current status of permission, certification, and reporting in 2023. In the case of pharmaceuticals, a total of 1,488 items were approved, including 1,300 finished medicines, 49 raw materials medicines, and 139 herbal medicines. In the case of finished drugs, 884 specialized drugs (68%) and 416 general drugs were approved. Last year, as the permission for rare drugs continued to increase, there were many permissions for diabetes treatments among all finished drugs. However, since the implementation of the 1+3 system, the permission for generic drugs has been steadily decreasing.
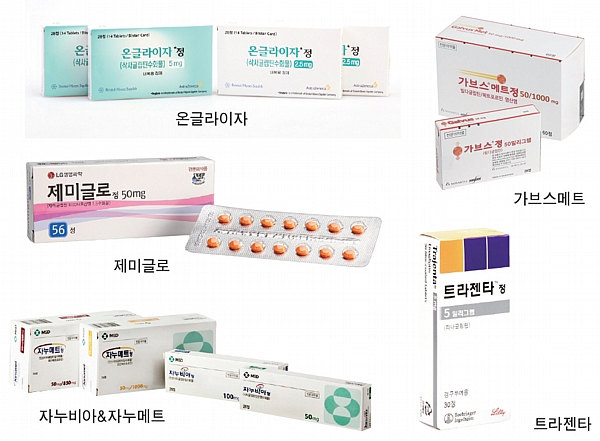
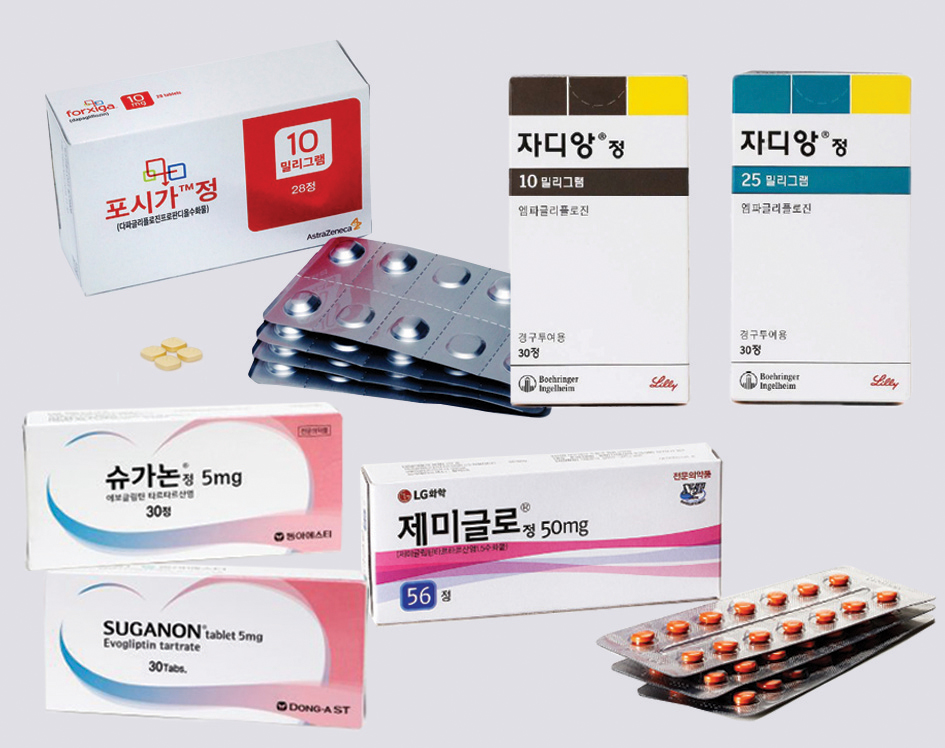
According to all carpenters of finished medicine licenses, diabetes treatments were the most approved in a total of 462 items. This is more than 30 percent of all finished drugs. It was followed by antipyretic, analgesic, and anti-inflammatory drugs (120 items), other vitamins (87 items), and blood pressure-lowering drugs (77 items). According to the status of chemical and pharmaceutical licenses approved last year, 29 new drugs, 22 rare drugs, 390 data submitted drugs (including 15 improved new drugs), and 2 raw materials drugs. The number of new drugs is 29 (5 manufactured and 24 imported) which is 50% higher than that of 2022, and the top efficacy classification is for nervous system drugs (7), diabetes drugs, anti-malignant tumor drugs (6 each), circulatory and blood/body fluid drugs, respiratory and allergic drugs (3 each). A total of 26 ingredients and 37 items were allowed for rare medicines, up 4 ingredients and 7 items from last year. The figure is about three times higher than in 2019 (9 ingredients), but along with increased investment in the pharmaceutical industry, it seems that the government has actively supported the development of rare drugs. The highest percentage of the drugs submitted in the data (375 items) was 50.4% (189 items), followed by 24.5% (92 items), and 13.3% (50 items), respectively. According to the recently approved development type of improved new drug, the development of new complex drugs (pharmaceuticals containing more than two main ingredients in one product) was active in 2016-2017, and six slow-release drugs were recognized as improved new drugs in 2018. Last year, it was found that a total of 15 items were allowed as 14 composite products with new active ingredient composition and 1 item with improved usefulness were approved by developing products with different efficacy and effects. According to all carpenters of finished medicine licenses, diabetes treatments were the most approved in a total of 462 items. This is more than 30 percent of all finished drugs. It was followed by antipyretic, analgesic, and anti-inflammatory drugs (120 items), other vitamins (87 items), and blood pressure-lowering drugs (77 items). According to the status of chemical and pharmaceutical licenses approved last year, 29 new drugs, 22 rare drugs, 390 data submitted drugs (including 15 improved new drugs), and 2 raw materials drugs. The number of new drugs is 29 (5 manufactured and 24 imported) which is 50% higher than that of 2022, and the top efficacy classification is for nervous system drugs (7), diabetes drugs, anti-malignant tumor drugs (6 each), circulatory and blood/body fluid drugs, respiratory and allergic drugs (3 each). A total of 26 ingredients and 37 items were allowed for rare medicines, up 4 ingredients and 7 items from last year. The figure is about three times higher than in 2019 (9 ingredients), but along with increased investment in the pharmaceutical industry, it seems that the government has actively supported the development of rare drugs. The highest percentage of the drugs submitted in the data (375 items) was 50.4% (189 items), followed by 24.5% (92 items), and 13.3% (50 items), respectively. According to the recently approved development type of improved new drug, the development of new complex drugs (pharmaceuticals containing more than two main ingredients in one product) was active in 2016-2017, and six slow-release drugs were recognized as improved new drugs in 2018. Last year, it was found that a total of 15 items were allowed as 14 composite products with new active ingredient composition and 1 item with improved usefulness were approved by developing products with different efficacy and effects.
Last year, 802 items were approved and reported for generic drugs. Other drugs, such as generic drugs, accounted for the highest percentage. Of the drugs submitted, 15 were recognized as improved new drugs. Due to the implementation of the “1+3” system, which allows the same clinical trial data to be used only three times from July 2021, the number of applications for permission has decreased sharply by 2022, and 2023 is similar to 2022. A total of 929 quasi-drugs were approved and reported. The main characteristic is that the development of quasi-drugs related to daily life such as sanitary napkins, bandages, and toothpaste has been activated due to the reduction in mask permission and reporting due to the easing of mandatory wearing of masks. A total of 7,065 medical devices have been approved, certified, and reported, ▲ Advanced medical devices such as artificial intelligence-based medical devices and surgical rehabilitation robots, ▲ Medical device software items are on the rise ▲ Digital therapy devices and heavy particle therapy devices have appeared. #Ministry of Food and Drug Safety 2023 Drug License Report #Number of Drug License Items Last year, 802 items were approved and reported for generic drugs. Other drugs, such as generic drugs, accounted for the highest percentage. Of the drugs submitted, 15 were recognized as improved new drugs. Due to the implementation of the “1+3” system, which allows the same clinical trial data to be used only three times from July 2021, the number of applications for permission has decreased sharply by 2022, and 2023 is similar to 2022. A total of 929 quasi-drugs were approved and reported. The main characteristic is that the development of quasi-drugs related to daily life such as sanitary napkins, bandages, and toothpaste has been activated due to the reduction in mask permission and reporting due to the easing of mandatory wearing of masks. A total of 7,065 medical devices have been approved, certified, and reported, ▲ Advanced medical devices such as artificial intelligence-based medical devices and surgical rehabilitation robots, ▲ Medical device software items are on the rise ▲ Digital therapy devices and heavy particle therapy devices have appeared. #Ministry of Food and Drug Safety 2023 Drug License Report #Number of Drug License Items